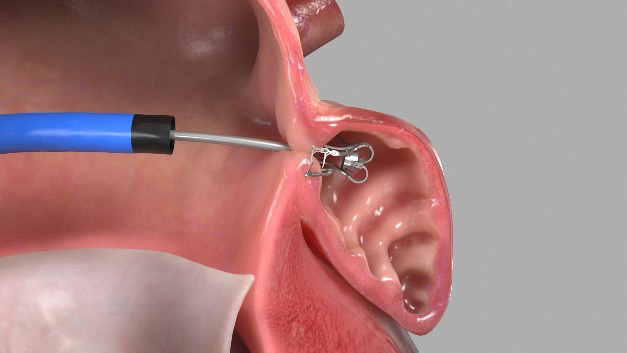
Endomatic develops a break through endovascular Left Atrial Appendage (“LAA”) closure device to prevent stroke.
Since a clot formation in the LAA is the primary cause for stroke in Atrial Fibralation patients, the interventional cardiologists and electrophysiologists routinely perform procedures which occlude the LAA with an implantable device.
As the implant come in contact with the heart’s flowing blood it may lead to device related thrombus (DRT), which is the clinicians highest concern, since it means that such procedures actively induce the stroke risk.
Endomatic patented technology expects to eliminate the risk of DRT due to its non-thrombogenic nature.
The company is endorsed with the top clinicians, including a world leader practitioner who became the head of its SAB and an investor.